 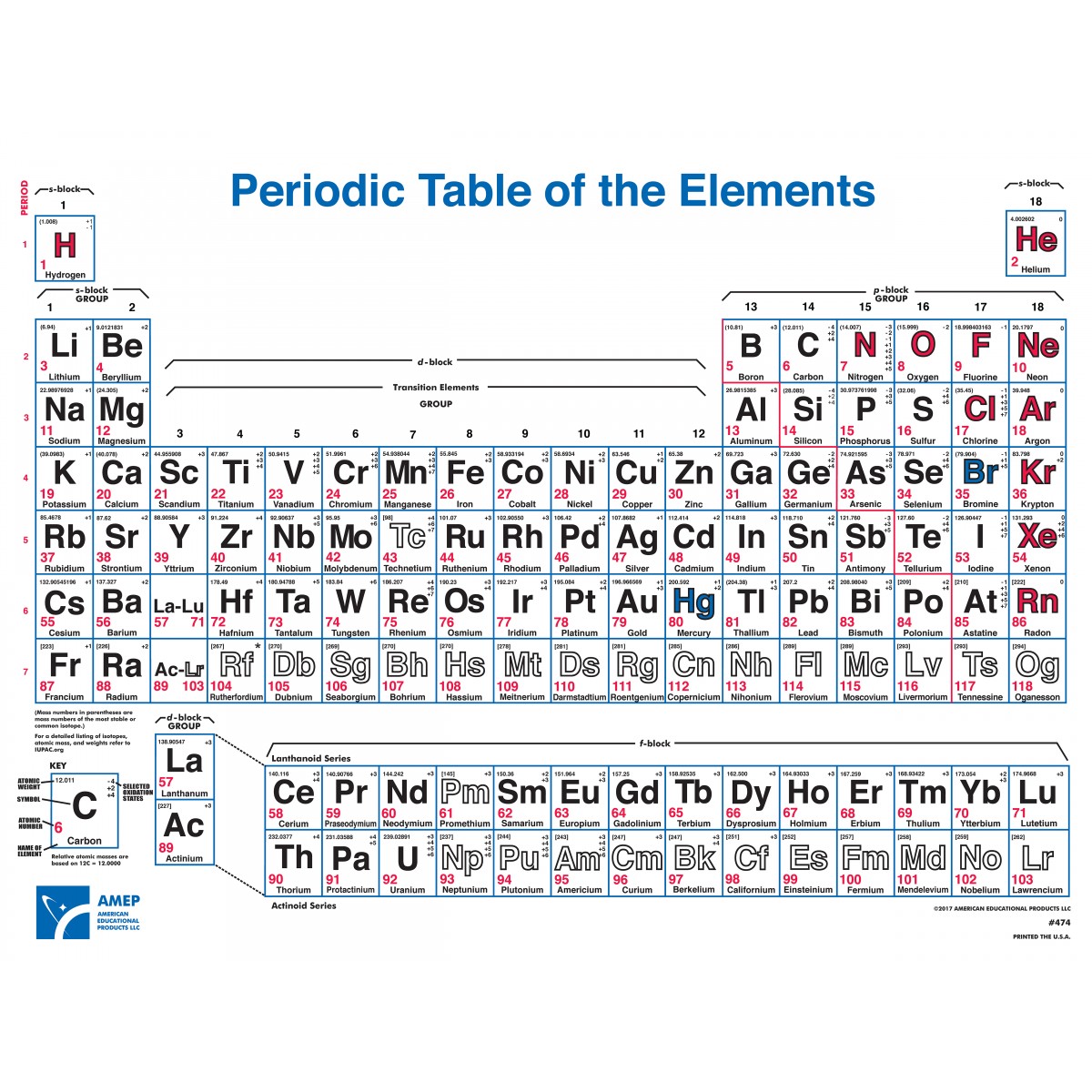
The characteristic properties of metals and nonmetals are quite distinct, as shown in the table below. Specialized subcategories such as the refractory metals and the noble metals also exist. From left to right in the periodic table, these categories include the highly reactive alkali metals the less-reactive alkaline earth metals, lanthanides, and radioactive actinides the archetypal transition metals and the physically and chemically weak post-transition metals. Metals comprise the large majority of the elements, and can be subdivided into several different categories. Au, Pt), or have nonmetallic structures ( Mn and Ga are structurally analogous to, respectively, white P and I). Ti, Re), or are noble (hard to oxidise, e.g. W, Nb), are liquids at or near room temperature (e.g. 
Be, Al) or very high melting points (e.g. Some metals appear coloured ( Cu, Cs, Au), have low densities (e.g. Most metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and electronegativities, and are found naturally in combined states. Metals appear lustrous (beneath any patina) form mixtures ( alloys) when combined with other metals tend to lose or share electrons when they react with other substances and each forms at least one predominantly basic oxide. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
